Q.E.D. Alcohol Saliva Test (CLIA Waived) : (Box of 10)
$68.75
The Q.E.D. was one of the first devices to be approved by the U.S. Department of Transportation (DOT) as an Alcohol Screening Device. DOT approval means that the Q.E.D. reliably and accurately detects alcohol at the 0.02 concentration or higher, and does not give false positive readings when no alcohol is present. DOT approval allows users to use the Q.E.D. for screening tests required by Dept. of Transportation regulations.
Semi-quantitative Results
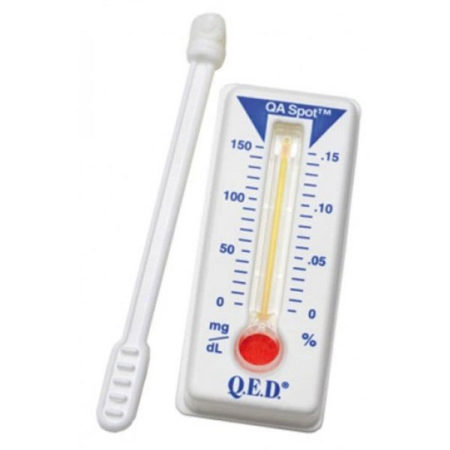
Because the proportion of alcohol in saliva is directly related to the proportion of alcohol in blood, the Q.E.D. device can accurately measure intoxication levels. Alcohol in the saliva creates a column of vivid reddish-purple color. The user reads the device at the upper limit of the color using the scale, exactly like reading a thermometer. If no alcohol is present no color develops.
Accurate as Blood Alcohol Test
The Q.E.D. device virtually duplicates blood alcohol results. Studies have demonstrated a .98 correlation (1.00 is a perfect correlation) between saliva test results with the Q.E.D. and laboratory blood alcohol results.
Description
– Quantitative results in 2-5 minutes.
– Reads just like a thermometer.
– Long shelf life
– Built-in quality control spot.
– CLIA waived, FDA 510K, DOT Cleared.
– High correlation to blood analysis.
– Non-invasive.
– DOT Approved as Alcohol Screening Device.
The Q.E.D. was one of the first devices to be approved by the U.S. Department of Transportation (DOT) as an Alcohol Screening Device. DOT approval means that the Q.E.D. reliably and accurately detects alcohol at the 0.02 concentration or higher, and does not give false positive readings when no alcohol is present. DOT approval allows users to use the Q.E.D. for screening tests required by Dept. of Transportation regulations.
Semi-quantitative Results
Because the proportion of alcohol in saliva is directly related to the proportion of alcohol in blood, the Q.E.D. device can accurately measure intoxication levels. Alcohol in the saliva creates a column of vivid reddish-purple color. The user reads the device at the upper limit of the color using the scale, exactly like reading a thermometer. If no alcohol is present no color develops.
Accurate as Blood Alcohol Test
The Q.E.D. device virtually duplicates blood alcohol results. Studies have demonstrated a .98 correlation (1.00 is a perfect correlation) between saliva test results with the Q.E.D. and laboratory blood alcohol results.
Instructions:
1. Actively swab around the cheeks, gums, and under the tongue for 30-60 seconds until the cotton swab is thoroughly saturated.
2. Place the Q.E.D.® test on a flat surface. Gently twist the collector into the entry port. Apply gentle steady pressure until the pink fluid passes the QA Spot at the end of the device.
3. Interpreting test results: Allow two (2) minutes for the Q.E.D.® A150 test to develop. The QA spot must be dark purple to indicate a valid test. If the same color forms a bar within the measurement scale, read the highest point. Ignore bubbles.
Additional information
| Weight | 1 lbs |
|---|